Alopecia Areata Market Summary
Alopecia Areata Insights and Trends
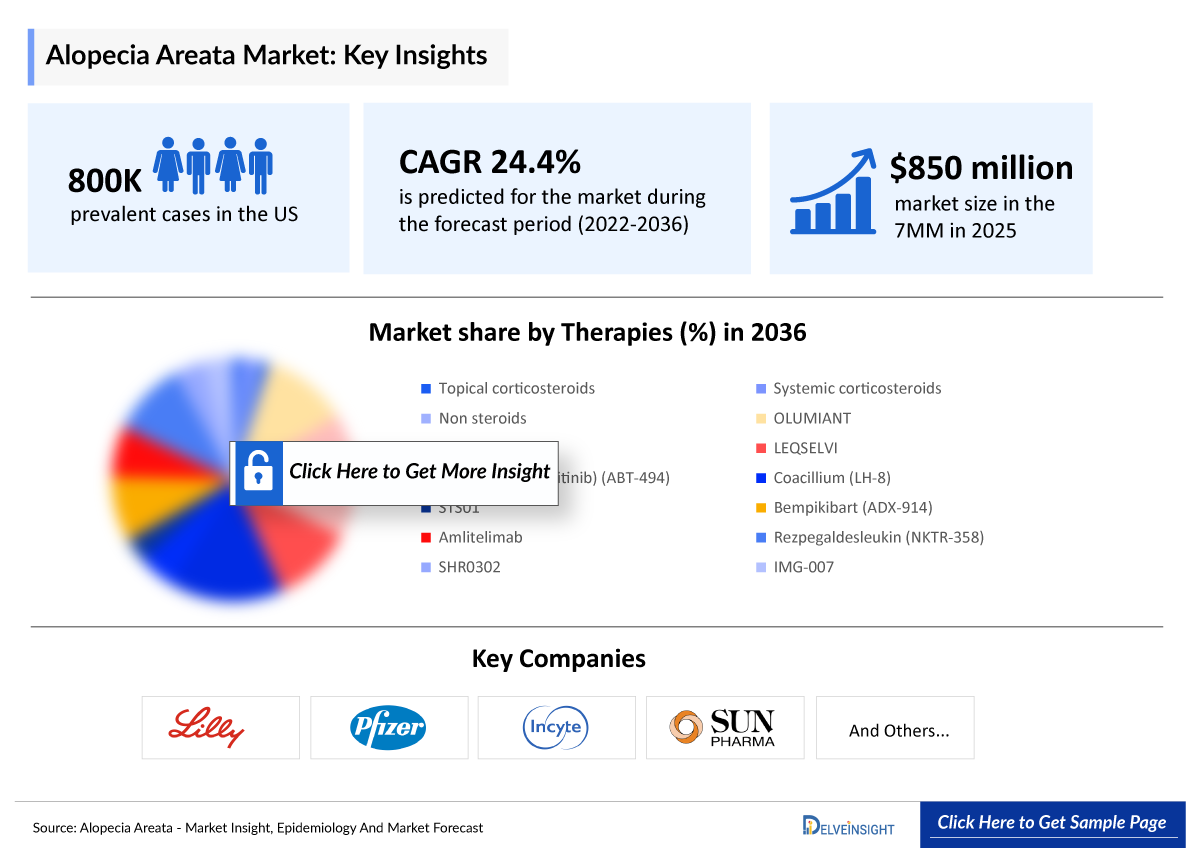
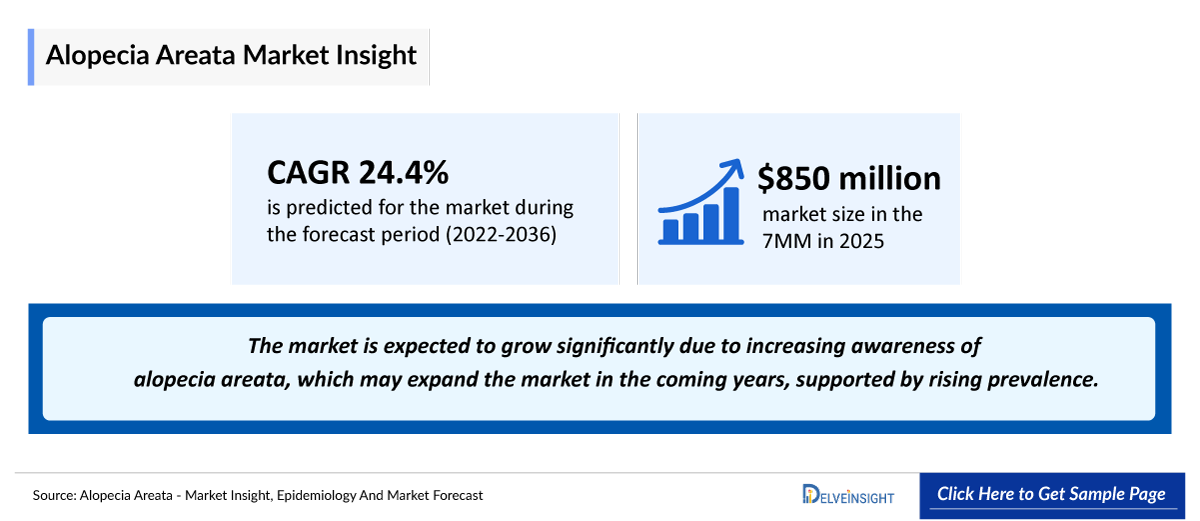
- According to DelveInsight’s analysis, alopecia areata market size was nearly USD 850 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Alopecia areata reported approximately 800,000 prevalent cases and around 630,000 diagnosed prevalent cases in the US.
- The current treatment landscape for alopecia areata is limited, with only three approved drugs available. Baricitinib (OLUMIANT), a JAK inhibitor developed by Eli Lilly and Incyte Corporation, was approved in 2022 for use in the US, EU4, the UK, and Japan. It marks a significant milestone as the first JAK inhibitor to gain approval for alopecia areata, targeting specific immune pathways involved in hair loss.
- Reliance on a single therapeutic class limits treatment diversity, as only JAK inhibitors are currently approved for alopecia areata, restricting options for patients who are non-responsive or unsuitable. Safety concerns associated with JAK inhibitors include increased risk of infections, malignancies, cardiovascular events, thrombosis, and laboratory abnormalities, limiting long-term use and reducing physician and patient acceptance in chronic treatment settings.
- Emerging therapies for alopecia areata include upadacitinib, coacillium, rezpegaldesleukin, and STS01, targeting immune pathways beyond JAK inhibition, focusing on immune regulation, tolerance restoration, and improving long-term safety and sustained hair regrowth outcomes.
- Despite the availability of various therapeutic options for alopecia areata, including approved and off-label therapies, challenges such as high costs and limited accessibility to current treatments persist. These issues underscore the necessity for continued innovation and approaches to managing the condition. Furthermore, comprehending treatment patterns is crucial to refining healthcare practices and advancing the treatment landscape for alopecia areata.
- Various pharmaceutical companies are working in this direction, upadacitinib (RINVOQ), also known as ABT-494, is developed by AbbVie, as an oral JAK1 selective inhibitor currently undergoing a Phase III clinical trial for severe alopecia areata. The trial, which is recruiting participants, is expected to be completed by March 2028. RINVOQ is a small molecule therapy targeting immune pathways involved in hair loss.
Alopecia Areata Market size and forecast
- 2025 Alopecia Areata Market Size in the 7MM: ~USD 850 million
- Alopecia Areata Growth Rate (2022-2036): 24.4% CAGR (Compound Annual Growth Rate)
DelveInsight’s “Alopecia Areata – Market Insights, Epidemiology, and Market Forecast – 2036” report delivers an in-depth understanding of alopecia areata, historical and forecasted epidemiology, as well as the alopecia areata market trends in the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
The alopecia areata market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, alopecia areata patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in alopecia areata and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Alopecia Areata Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Alopecia areata Market CAGR (Forecast period) |
24.4% (2022-2036) |
|
Alopecia Areata Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Alopecia Areata Companies |
|
|
Alopecia Areata Therapies |
|
|
Alopecia Areata Market Segmentation |
Segmented by
|
|
Alopecia Areata Market Analysis |
|
Alopecia Areata Understanding and Treatment Algorithm
Alopecia Areata Overview and Diagnosis
Alopecia areata is a common autoimmune skin disease, causing hair loss on the scalp, face, and sometimes on other areas of the body, affecting people of all ages, both sexes and all ethnic groups can develop alopecia areata. It is one of many recognized forms of alopecia; alopecia areata is the second most common form after androgenetic alopecia. It often first appears during childhood and can be different for everyone who has it. Visible symptoms include small, coin-sized, round patches of baldness on the scalp, although hair elsewhere, such as the beard, eyebrows, eyelashes, body, and limbs, can be affected. In some people, larger areas are affected, and occasionally, it can involve the whole scalp (alopecia totalis) or even the entire body and scalp.
The evaluation of alopecia areata includes a comprehensive medical and family history along with a detailed examination of the scalp, face, body, and nails, supported by dermatoscopy (trichoscopy) and a hair pull test. When diagnosis remains uncertain, additional investigations such as scalp biopsy, fungal culture, or serological tests for autoimmune or infectious diseases may be required. Trichoscopy serves as a noninvasive and essential diagnostic tool to differentiate scarring from nonscarring alopecia, confirm diagnosis, and assess disease activity, with characteristic findings including yellow dots, black dots, broken hairs, exclamation mark hairs, vellus hairs, and less commonly upright, tapered, pigtail hairs, and Pohl-Pinkus constrictions.
Further details related to country-based variations are provided in the report…
Current Alopecia Areata Treatment Landscape
The alopecia areata treatment landscape in is rapidly evolving, with various therapies targeting immune dysregulation and promoting hair regrowth. Topical corticosteroids are commonly used first-line to suppress localized inflammation and T-cell activity in hair follicles. However, their long-term use is limited by side effects like skin thinning.For more severe cases, systemic corticosteroids, administered orally or intralesionally, reduce inflammation and promote regrowth, though prolonged use can lead to side effects such as weight gain, hypertension, and adrenal suppression.
The alopecia areata treatment landscape in the currently includes three FDA-approved therapies, all belonging to the JAK inhibitor class. OLUMIANT, developed by Eli Lilly and Company and Incyte Corporation, was the first approved in 2022, marking a major breakthrough in targeted treatment. LITFULO, developed by Pfizer, followed in 2023 as a selective JAK3 and TEC kinase inhibitor, expanding treatment options, including for adolescents. Most recently, LEQSELVI by Sun Pharmaceutical Industries was launched in 2025 as a JAK1/2 inhibitor for adults with severe disease, further strengthening the targeted therapy landscape.
Further details related to country-based variations are provided in the report.
Alopecia Areata Unmet Needs
The section “unmet needs of alopecia areata” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Complex etiology and unpredictable disease nature
- Wigs as cranial prostheses
- Limitations of current therapies
- Need to increase awareness
- Access to treatments and cost barriers
- and others…..
Note: Comprehensive unmet needs insights in alopecia areata and their strategic implications are provided in the full report.
Alopecia Areata Epidemiology
Key Findings from Alopecia Areata Epidemiological Analysis and Forecast
- In 2025, the US accounted for the highest prevalent cases of alopecia areata with approximately 800,000 cases, which are expected to increase by 2036.
- In 2025, the UK reported the highest number of diagnosed prevalent cases of alopecia areata among EU4 and the UK, with approximately 330,000 cases. Spain followed with around 180,000 cases, while Italy recorded the lowest diagnosed prevalence, with nearly 100,000 cases.
- In 2025, Japan reported approximately 170,000 thousand diagnosed prevalent cases of alopecia areata in females and nearly 110,000 cases in males, with numbers projected to rise by 2036.
- In 2025, females accounted for 63% of alopecia areata cases in EU4 and the UK, while males represented 37%. These gender-specific patterns are expected to grow, with overall prevalence projected to increase by 2036.
- In 2025, around 14% of alopecia areata cases in EU4 and the UK were mild, 45% moderate, and 41% severe. These severity patterns are expected to continue, with overall prevalence anticipated to increase by 2036.
Alopecia Areata Drug Analysis & Competitive Landscape
The alopecia areata drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I-III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the alopecia areata treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the alopecia areata therapeutics market.
Approved Therapies for Alopecia Areata
Deuruxolitinib (LEQSELVI): Sun Pharmaceuticals
Deuruxolitinib, an FDA-approved treatment for severe alopecia areata in adults, functions as an inhibitor of JAK1 and JAK2. Developed by Sun Pharmaceutical Industries, this therapy targets pathways implicated in the autoimmune processes responsible for hair loss in alopecia areata. Its efficacy was demonstrated in clinical trials such as THRIVE-AA1 and THRIVE-AA2, which highlighted significant improvements in scalp hair regrowth. Despite its benefits, LEQSELVI carries risks of severe adverse effects, including infections, malignancies, and thrombosis, requiring cautious use, particularly in at-risk populations.
- The US FDA approved Sun Pharma's deuruxolitinib in July 2024 to treat severe alopecia areata. Despite the fact that patent litigation initially caused a delay in its introduction, the injunction was lifted in April 2025 and the disagreement was later settled, allowing for its commercial launch in the US in 2025.
Alopecia Areata Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Deuruxolitinib (LEQSELVI) |
Sun Pharmaceuticals |
Alopecia areata |
Small molecule |
JAK1 inhibitors; JAK2 inhibitors |
Oral |
US: 2024 |
|
Ritlecitinib (LITFULO)
|
Pfizer |
Alopecia areata |
Small molecule |
JAK3 inhibitors; TEC kinases inhibitors |
Oral |
US: 2023 EU: 2023 JP: 2023 |
Note: Detailed marketed therapies assessment will be provided in the final report.
Alopecia Areata Pipeline Analysis
STS-01: Soterios
Soterios’ lead program, STS-01, has completed a Phase II trial targeting alopecia areata. Designed as a non-steroidal topical therapy, it leverages a mechanism with an established dermatological safety profile and demonstrated efficacy. Unlike JAK inhibitors, which face limitations due to side effects and cost, STS-01 offers an effective, safe, and cosmetically appealing alternative.
- In May 2024, Soterios Pharma reported promising Phase II trial results for STS-01 in mild-to-moderate alopecia areata. STS-01 was well-tolerated with no significant adverse events, and full results are anticipated to be presented later.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Upadacitinib (RINVOQ) |
AbbVie |
III |
Severe alopecia areata |
Oral |
JAK1 selective inhibitor |
2027 |
|
Coacillium (LH-8) |
Legacy Healthcare |
III |
Moderate to severe alopecia areata |
Cutaneous solution |
Immuno-inflammatory modulator |
Information is available in the full report |
|
Rezpegaldesleukin (NKTR-358) |
Nektar Therapeutics |
II |
Severe-to-very severe alopecia areata |
Subcutaneous |
IL-2 T regulatory cell stimulator |
Information is available in the full report |
|
STS01 |
Soterios |
II |
Mild-moderate alopecia areata |
Topical cream |
Cytokine modulator |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report.
Alopecia Areata Key Players, Market Leaders and Emerging Companies
- Eli Lilly and Company/ Incyte Corporation
- Pfizer
- Sun Pharmaceuticals
- AbbVie
- Legacy Healthcare
- Nektar Therapeutics and others
Alopecia Areata Drug Updates
- In January 2026, Legacy Healthcare reported plans to advance its topical solution Coacillium (LH-8) into an international Phase III clinical trial for Alopecia Areata, following clearance from the US FDA.
- In August 2025, AbbVie reported positive topline results from second Phase III UP-AA trial evaluating upadacitinib (RINVOQ) for alopecia areata.
- In July 2024, Sun Pharmaceuticals reported that the US FDA approved Deuruxolitinib (LEQSELVI), a once-daily oral treatment, for severe alopecia areata.
Alopecia Areata Market Outlook
The treatment landscape for alopecia areata has evolved to incorporate a comprehensive approach, combining medications with supportive care. The market offers a range of therapies, including corticosteroids, immunosuppressants, immunomodulators, and topical sensitizers or irritants. Corticosteroids remain central for their anti-inflammatory properties, reducing immune attacks on hair follicles, while immunosuppressants and immunomodulators are used in more severe or resistant cases to regulate the immune system and prevent further hair loss.
Topical sensitization therapies, like diphencyprone, induce a mild allergic reaction to divert the immune response from hair follicles. Similarly, topical irritation therapies trigger irritation to stimulate hair regrowth. Minoxidil, a vasodilator, promotes blood flow to hair follicles and is often used off-label for alopecia areata. Dithranol, less commonly used, creates controlled inflammation to alter immune responses and encourage regrowth, primarily for patchy alopecia areata as part of sensitization therapy.
Some drugs are currently approved and available for the treatment of alopecia areata, reflecting the focus on immune modulation therapies. Key options include OLUMIANT, a small molecule co-developed by Eli Lilly and Company and Incyte Corporation, and LITFULO, a JAK3 and TEC kinase inhibitors offered by Pfizer. LEQSELVI, JAK1 and JAK2 inhibitors developed by Sun Pharmaceuticals.
There is a significant need for new treatments for alopecia areata, as existing therapies often fall short of fully managing the condition. Innovative drug development could provide more effective options for patients. Several promising drugs are currently in the pipeline, including Upadacitinib (RINVOQ), coacillium (LH-8), and STS01, among others.
- In 2025, the alopecia areata market size in the US was around USD 500 million, accounting for 53% of the total market. This figure is expected to grow significantly with the introduction of emerging therapies.
- The total market size of EU4 and the UK was estimated to be approximately USD 300 million in 2025, which was nearly 39% of the total market revenue for the 7MM.
- Among EU4 and the UK, the UK accounted for the highest market share with approximately USD 90 million in 2025, followed by Germany with approximately USD 55 million in the respective year, and France, capturing the least market with nearly USD 40 million in 2025.
- In 2025, the total market size of alopecia areata was approximately USD 60 million in Japan which is anticipated to increase during the forecast period (2026–2036).
Drug Class/Insights into Leading Emerging Therapies in Alopecia Areata (2022–2036 Forecast)
Current therapies for alopecia areata aim to immunosuppress or immunomodulate the activity of the disease, with generally unsatisfying responses and high relapse rates, especially in more severe cases. Due to the unpredictable course of the disease and the spontaneous remissions often observed within the first year, therapy efficacy is difficult to estimate. Additionally, the available therapeutic options do not influence the long-term course of the disease, and thus, an urgent need remains for novel, more effective therapies.
- Intralesional Corticosteroids
Effective for small, localized areas, typically with triamcinolone acetonide. Injections are often spaced out, and concentrations vary based on the site of treatment (scalp vs. face). Risks include skin atrophy, hypopigmentation, and rare side effects like cataracts when used near the eyes.
- Potent Topical Corticosteroids
Used as first-line treatment, especially in children or patients who avoid injections. Evidence of efficacy is limited, with mixed results from randomized trials. Side effects can include skin irritation, folliculitis, and potential adrenal suppression.
- Systemic Glucocorticoids
Oral prednisone may promote regrowth in patchy alopecia areata but may be less effective in alopecia totalis or universalis. Long-term use is associated with risks, including recurrence of the disease after cessation.
- Topical Immunotherapy
Often the preferred first-line treatment for extensive chronic alopecia areata involving allergens like Diphenylcyclopropenone (DPCP) or Squaric Acid Dibutylester (SADBE). Efficacy rates for complete regrowth can vary, with patchy alopecia areata responding better than totalis or universalis.
- Topical Anthralin
Historical use for psoriasis, with some evidence suggesting efficacy in alopecia areata when applied for short periods. Combination therapy with DPCP may be more effective than monotherapy.
- Minoxidil
Evidence for effectiveness is mixed, but it is commonly used to enhance regrowth in patchy alopecia areata. 5% minoxidil is more effective than 1%, though it’s not recommended for alopecia totalis/universalis.
- Biologic Drugs
Some biologic agents (e.g., TNF-α inhibitors) have not shown expected results, and some may even exacerbate alopecia areata. JAK inhibitors, particularly tofacitinib, and ruxolitinib, have shown promise in treating both patchy alopecia areata and alopecia totalis/universalis, with good clinical responses within a few months.
- Systemic Immunosuppressive Agents
Methotrexate and azathioprine can be useful, particularly when combined with corticosteroids. Side effects may include liver toxicity, bone marrow suppression, and gastrointestinal issues.
- Miscellaneous Treatments
Aromatherapy and bexarotene show limited or inconclusive efficacy.Laser therapies (e.g., excimer laser) and platelet-rich plasma) have mixed results, with PRP showing promise for chronic alopecia areata.
Alopecia Areata Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the alopecia areata drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The treatment landscape for alopecia areata in the US has seen advancements, particularly with therapies targeting immune dysregulation. Despite this progress, a significant need remains, as existing treatments often fall short in addressing severe or refractory cases. Corticosteroids, though widely used, are associated with limited efficacy in promoting sustained hair regrowth and carry risks of adverse effects, particularly with prolonged use. Similarly, systemic therapies may offer short-term benefits but are constrained by their side effect profiles and the lack of durable remission in many patients.
The development of therapies with innovative mechanisms of action represents a significant advancement in treating alopecia areata. These treatments specifically target pathways involved in the autoimmune destruction of hair follicles, providing a more focused approach and promising improved effectiveness and outcomes for patients facing this challenging condition.
AbbVie is advancing Upadacitinib as a Phase III candidate targeting patients with severe alopecia areata. Based on the company’s latest presentation, regulatory approvals for this indication are anticipated around 2027.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Therapies in Alopecia Areata
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
EU4 and the UK
EU4 and the UK Reimbursement of Therapies Approved for Alopecia Areata | |
|
Drug/Therapy |
Indication |
|
Ritlecitinib (LITFULO) |
The NICE has recommended ritlecitinib as a treatment option for severe alopecia areata in individuals aged 12 and older in the UK |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Alopecia Areata Therapies Price Scenario & Trends
Pricing and analogue assessment of alopecia areata therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Olumiant, an approved therapy for alopecia areata, is associated with a relatively high annual treatment cost in the United States, estimated at approximately USD 30,000. This pricing reflects its status as a targeted oral JAK inhibitor and may pose affordability challenges, particularly in long-term disease management settings.
Industry Experts and Physician Views for Alopecia Areata
To keep up with alopecia areata market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the alopecia areata emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in alopecia areata, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the Brigham and Women's Hospital, Atrium Health Wake Forest Baptist, Carver College of Medicine , Centre hospitalier universitaire de Nantes etc. were contacted.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Early detection is pivotal in improving patient outcomes and enhancing healthcare efficiency. It enables access to a broader range of treatment options, including less aggressive therapies, which greatly enhance a patient’s quality of life. Timely diagnosis is strongly associated with higher survival rates, as it prevents the disease from advancing to more severe stages.” |
|
Germany |
“Recent studies have highlighted the critical role of CD8+NKG2D+ T cells as key effectors in the pathogenesis of alopecia areata. Both mouse and human studies have shown gene expression profiles indicative of cytotoxic T cell infiltration, heightened IFN-γ responses, and upregulation of γ-chain cytokines like IL-2 and IL-15, which support the activation and persistence of these effector T cells. Additionally, Vδ1+ T cells with pro-inflammatory properties were significantly enriched in the supra bulbar and bulbar regions of affected hair follicles, providing further insight into the immunological mechanisms driving this disease.” |
Their opinion helps understand and validate current and emerging alopecia areata therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in alopecia areata.
Qualitative Analysis: SWOT and Attribute Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and attribute analysis.
In the SWOT analysis of alopecia areata, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provide attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Alopecia Areata Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of alopecia areata, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the alopecia areata market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM alopecia areata market.
Alopecia Areata Market Report Insights
- Alopecia Areata Patient Population Forecast
- Alopecia Areata Therapeutics Market Size
- Alopecia Areata Pipeline Analysis
- Alopecia Areata Market Size and Trends
- Alopecia Areata Market Opportunity (Current and Forecasted)
Alopecia Areata Market Report Key Strengths
- Epidemiology‑based (Epi‑based) bottom‑up forecasting
- Artificial Intelligence (AI) - enabled market research report
- 11-year forecast
- Alopecia Areata Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Alopecia Areata Treatment Addressable Market (TAM)
- Alopecia Areata Competitive Landscape
- Alopecia Areata Major Companies Insights
- Alopecia Areata Price Trends and Analogue Assessment
- Alopecia Areata Therapies Drug Adoption/Uptake
- Alopecia Areata Therapies Peak Patient Share analysis
Alopecia Areata Market Report Assessment
- Alopecia Areata Current Treatment Practices
- Alopecia Areata Unmet Needs
- Alopecia Areata Clinical Development Analysis
- Alopecia Areata Emerging Drugs Product Profiles
- Alopecia Areata Market Attractiveness
- Alopecia Areata Qualitative Analysis (SWOT and attribute analysis)
Key Questions Answered in the Alopecia Areata Market Report
Market Insights
- What was the alopecia areata market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of alopecia areata?
- What are the disease risks, burdens, and unmet needs of alopecia areata? What will be the growth opportunities across the 7MM concerning the patient population with alopecia areata?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of alopecia areata? What are the current guidelines for treating alopecia areata in the US, Europe, and Japan?
Reasons to Buy Alopecia Areata Market Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the alopecia areata market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) ‑ enabled report summarize and simplify complex datasets with in the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.