Gene therapy
Jul 04, 2023
First Gene Therapy for Severe Hemophilia A; FDA Approves CellTrans’s Type 1 Diabetes Cellular Therapy; FDA Approve Amneal’s Parkinson’s Drug Over Safety Concerns; NLS Pharmaceutics’s Phase III Clinical Trials of Quilience; Genenta Science’s Temferon for the Treatment of Glioma; FDA Fast Track Designation to GSK’s Gonorrhea Drug
FDA Approves First Gene Therapy for Severe Hemophilia A BioMarin Pharmaceutical Inc., a global biotechnology company dedicated to transforming lives through genetic discovery, announced that the US Food and Drug Administration (FDA) has approved ROCTAVIAN (valoctocogene roxaparvovec-rvox) gene therapy for the tr...
Read More...
Jun 09, 2023
Cell and Gene Therapies in Rare Disorders: From Rarity to Recovery
The cell and gene therapy market has seen a revolutionary transition in recent years, with advancements in scientific research and novel methods of treatment driving a rise in development activities. This has resulted in an increase in the number of cell and gene therapy choices available to patients suffering from...
Read More...
May 16, 2023
Sarepta Therapeutics’s SRP-9001 Gene Therapy; FDA Approves Astellas’ VEOZAH; FDA Orphan Drug Designation and Rare Pediatric Disease Designation to SiSaf’s siRNA Therapy SIS-101-ADO; FDA Grants Fast Track Designation to IMPT-314; FDA Approves First Drug for Agitation in People With Alzheimer’s Disease; FDA Accepted the CytoAgents’ IND Application for CTO1681
Sarepta Therapeutics Announces Positive Vote from U.S. FDA Advisory Committee Meeting for SRP-9001 Gene Therapy Sarepta Therapeutics, Inc., a pioneer in precision genetic medicine for rare diseases, announced that the FDA's Cellular, Tissue, and Gene Therapies Advisory Committee (CTGTAC) voted 8 to 6 in favor of...
Read More...
May 12, 2023
Gene Therapy in Rare Disorders: Acceptance in Europe Faces Challenges
By bringing life-changing benefits to people with rare diseases and cancers, cell and gene therapies are reshaping the field of medicine. These treatments have recently gained tremendous publicity for meeting long-standing unmet needs and being extremely costly. But high production costs are limiting patient access...
Read More...
May 08, 2023
Watershed Moment for Cell Therapies and Complicated Journey of Gene Therapies in Japan
In recent years, the development of cell and gene therapies has provided a new avenue for the treatment of rare disorders. This article will discuss the current and future scenarios of cell and gene therapies in rare disorders with cancer indications as an exception, in Japan. Cell therapy is a treatment in which l...
Read More...
May 01, 2023
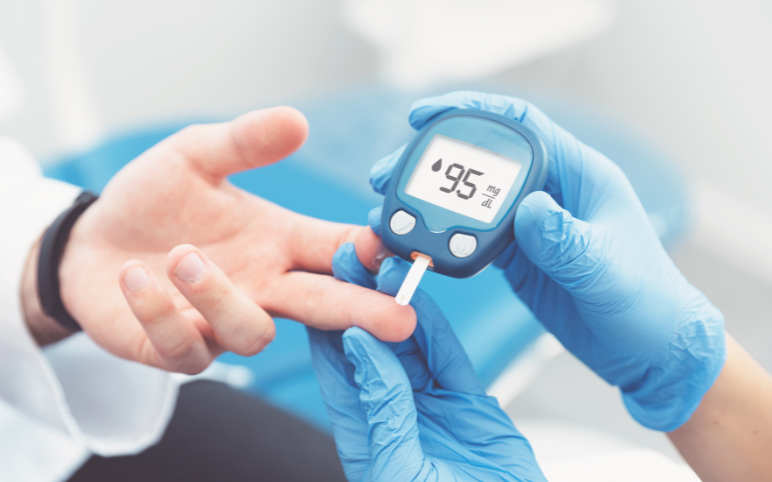
Cell and Gene Therapies for Diabetes Treatment: A Permanent Cure for Patients?
Diabetes is the 8th largest cause of death in the United States (although its prevalence may be underreported). Diabetes affects more than 37 million people in the United States, and 1 in every 5 are unaware of their condition. Over 96 million US adults—more than one-third—have prediabetes, and more than 8 out of 1...
Read More...
Apr 14, 2023
Vertex/CRISPR’s Gene-editing Therapy exa-cel: Inch Ahead of Rival
Vertex Pharma and CRISPR Therapeutics are the first companies to seek FDA clearance for a gene-editing therapy. Vertex Pharmaceuticals and CRISPR Therapeutics have gotten closer to introducing exagamglogene autotemcel (exa-cel), a one-time treatment for sickle cell disease (SCD) and transfusion-dependent beta-...
Read More...
Jan 24, 2023
BeiGene’s Brukinsa Approval; FDA Approval to Seagen’s TUKYSA; NICE Recommends Alnylam’s Amvuttra; FDA Approves Brenzavvy for Type 2 Diabetes; Roche’s Tecentriq to be Filed for Early-stage Liver Cancer; FDA Lifts Hold on Astellas’ Pompe Gene Therapy
FDA Approves BeiGene’s Brukinsa for CLL/SLL BeiGene's Brukinsa (zanubrutinib) for chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL) has been approved by the US Food and Drug Administration. CLL is a common type of leukemia, accounting for approximately 25% of all new cases each year. SLL is...
Read More...
Jan 03, 2023
Gilead Buys Out Rights to Cancer Therapy from Jounce; FDA Places Clinical Hold on Biogen’s Orelabrutinib; Pfizer Announces Phase 3 BENEGENE-2 Study Result; FDA Approves MediWound’s NexoBrid; UCB Announces FDA Acceptance of BLA Resubmission for Bimekizumab; FDA Approves TG Therapeutics’ Briumvi
Gilead Buys Out Rights to Cancer Therapy from Jounce for USD 67 Million Gilead Sciences must have liked what it saw in a two-year-old collaboration with Jounce Therapeutics for CCR8-targeting cancer immunotherapy because the company has just agreed to own the program fully. The drug in question, GS-1811 (formerl...
Read More...
Dec 20, 2022
Fourth FDA Approval for AbbVie’s Vraylar; FDA Approves Ferring’s Adstiladrin for NMIBC; Merck and Moderna’s mRNA Cancer Vaccine Trial; EMA Recommends the CSL’s Gene Therapy for Hemophilia B; CHMP Backs Amicus’ Pompe Disease Therapy; Takeda Announces the Phase 3 AURORA Study Result
AbbVie Secures Fourth FDA Approval for Vraylar AbbVie has received its fourth FDA approval for Vraylar, adding major depressive disorder (MDD) adjunctive therapy to a list that includes schizophrenia and manic and depressive episodes in bipolar disorder. According to AbbVie, the approval makes Vraylar (cariprazi...
Read More...









-Agonist.png)

