WET AMD
Oct 22, 2024
Gilead and Merck Announce Encouraging Phase II Results of Islatravir and Lenacapavir Combo; REGENXBIO Highlights Positive Data from Phase II ABBV-RGX-314 Wet AMD Study at AAO 2024; FDA Greenlights Astellas’ VYLOY for Advanced Gastric and GEJ Cancer Therapy; Merck’s CAPVAXIVE Vaccine Yields Strong Immune Response in Adults Vulnerable to Pneumococcal Disease; FDA Postpones LUMAKRAS Colorectal Cancer Verdict to Early 2025
Gilead and Merck Reports Phase II Data Showing that Switching to a Once-Weekly Oral Combination of Islatravir and Lenacapavir Maintained Viral Suppression in Adults Through 48 Weeks Gilead Sciences, Inc. and Merck have shared new findings from a Phase II clinical trial assessing the experimental combination of i...
Read More...
Jul 09, 2024
Lilly’s Morphic Acquisition; IDEAYA’s IDE397 Positive Phase II Trial Result; XPOVIO (selinexor) Approval in China; Roche to Reintroduce Susvimo in the US; Dupixent EU Approval
Lilly Strengthens IBD Treatment Portfolio with Morphic Acquisition Eli Lilly and Company and Morphic Holding, Inc. announced a definitive agreement for Lilly to acquire Morphic, a biopharmaceutical company developing oral integrin therapies for serious chronic diseases. Lilly will initiate a tender offer to acqu...
Read More...
Feb 06, 2024
4DMT Presents Data From Phase II PRISM Clinical Trial; Adaptimmune Announces FDA Acceptance of Biologics License Application for Afami-cel; Astellas Submits Supplemental New Drug Application in Japan for PADCEV with KEYTRUDA; AnaMar Announces US and EU Orphan Drug Designation for AM1476; Biosyngen Announces FDA Fast Track Designation for BST02; Acepodia Announces FDA Clearance of IND Application for ACE2016
4DMT Presents Positive Interim Data from Randomized Phase II PRISM Clinical Trial of Intravitreal 4D-150 Demonstrating Favorable Tolerability & Clinical Activity in Wet AMD 4D Molecular Therapeutics, a prominent company in the field of genetic medicines with a focus on harnessing the full potential of geneti...
Read More...
Oct 16, 2017
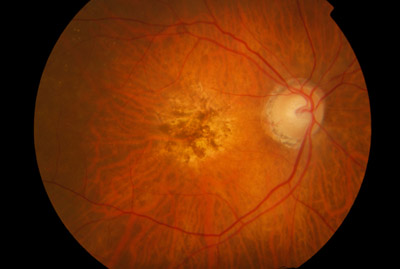
Wet AMD: A chronic eye condition with promising therapies
Wet AMD is a chronic eye disease that causes blurred vision or a blind spot in a person’s visual field. It is characterized by abnormal blood vessels that leak blood and fluids into the macula, a region of the eye that is responsible for sharp, central vision. Age-related macular degeneration (AMD) is a common eye c...
Read More...



-Agonist.png)

