Hepatitis C Market
Key Highlights
- Hepatitis C is a viral infection primarily affecting the liver, leading to acute and chronic liver disease if untreated. Globally, around 50 million people have chronic Hepatitis C, with approximately 1 million new infections annually. The WHO reported that in 2022, about 242,000 deaths were attributed to Hepatitis C, primarily due to cirrhosis and hepatocellular carcinoma.
- In 2023, Genotype 1 accounted for approximately 70% of genotype-specific diagnosed Hepatitis C cases in the United States, highlighting its prevalence and the need for targeted treatment strategies.
- Hepatitis C treatment has been transformed by direct-acting antivirals (DAAs) such as HARVONI and Epclusa from Gilead Sciences, and VIEKIRA PAK from AbbVie, which offer high cure rates and target various stages of the virus. AbbVie’s MAVYRET, introduced in 2017, further expanded treatment options, enhancing the global effort to eliminate Hepatitis C.
- In June 2024, the FDA granted marketing authorization to Cepheid for the Xpert HCV test and GeneXpert Xpress System, marking a significant advancement in Hepatitis C diagnostics by enabling a more rapid, test-and-treat approach compared to traditional multi-step testing.
- Despite advancements, there remains no effective vaccine for Hepatitis C, underscoring the need for continued research to improve treatment and diagnostics. The current pipeline is limited, highlighting an ongoing unmet need for innovative solutions in Hepatitis C management.
DelveInsight's “Hepatitis C – Market Insights, Epidemiology and Market Forecast – 2034” report delivers an in-depth understanding of hepatitis C, historical and forecasted epidemiology as well as the hepatitis C market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
Hepatitis C market report provides real-world prescription pattern analysis, emerging drugs, market share of individual therapies, and historical and forecasted 7MM Hepatitis C market size from 2020 to 2034. The report also covers current Hepatitis C treatment practices/algorithms and unmet medical needs to curate the best opportunities and assess the market’s underlying potential.
Geography Covered
- The United States
- EU4 (Germany, France, Italy, and Spain) and the United Kingdom
- Japan
| Study Period | 2020–2034 |
| Forecast Period | 2024–2034 |
| Geographies Covered | US, EU4 (Germany, France, Italy, and Spain) the UK, and Japan |
| Hepatitis C Epidemiology
| Segmented by:
|
| Hepatitis C key companies |
|
| Hepatitis C key therapies/drug |
|
| Hepatitis C Market | Segmented by:
|
| Analysis |
|
Hepatitis C Understanding and Treatment Algorithm
Hepatitis C Overview, Country-Specific Treatment Guidelines and Diagnosis
Hepatitis C is a serious viral infection that primarily targets the liver, leading to both acute and chronic illness, which can be life-threatening if left untreated. Transmitted through contact with infected blood, common modes of transmission include sharing needles, syringes, and undergoing unsafe medical procedures involving unscreened blood products. While symptoms like jaundice, fatigue, and abdominal pain can indicate the presence of the virus, many cases go undiagnosed due to the often asymptomatic nature of the infection in its early stages. Although there is no vaccine for hepatitis C, advancements in antiviral treatments offer effective management and the possibility of a cure
The Hepatitis C report provides an overview of Hepatitis C pathophysiology, diagnostic approaches, and detailed treatment algorithm along with a real-world scenario of a patient’s journey beginning from the first symptom, the time taken for diagnosis to the entire treatment process.
Further details related to country-based variations in diagnosis are provided in the report
Hepatitis C Treatment
Hepatitis C treatment has greatly advanced with the advent of direct-acting antivirals (DAAs), which can cure over 90% of cases. The standard regimen involves 8 to 24 weeks of oral DAA therapy, tailored to the patient's genotype, liver disease stage, and treatment history. These medications effectively disrupt the virus's replication process, leading to a sustained virologic response (SVR), where the virus remains undetectable 12 weeks post-treatment, signifying a cure. Supporting liver health through lifestyle changes, such as avoiding alcohol and maintaining a healthy weight, further enhances treatment outcomes.
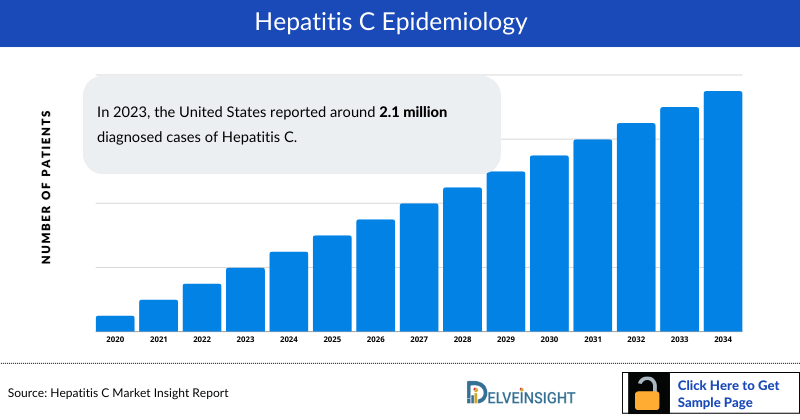
Hepatitis C Epidemiology
The Hepatitis C epidemiology chapter in the report provides historical as well as forecasted in the 7MM covering the United States, EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan from 2024 to 2034. The Hepatitis C epidemiology is segmented with detailed insights into total prevalent cases of Hepatitis C virus, total diagnosed cases of Hepatitis C virus, gender-specific cases of Hepatitis C virus, age-specific diagnosed cases of Hepatitis C virus, genotype-specific diagnosed cases of Hepatitis C virus, diagnosed cases of Hepatitis C virus by impact on liver, and treated cases of Hepatitis C virus
- In 2023, the United States reported around 2.1 million diagnosed cases of Hepatitis C.
- In the same year, males comprised approximately 60% of cases across the EU4 and the UK.
- In Japan, the 46-55 age group accounted for about 70% of Hepatitis C cases in 2023.
Hepatitis C Recent Developments
- In March 2025, following a successful End-of-Phase 2 meeting with the FDA in January 2025, Atea is launching its global Phase 3 program for hepatitis C, with patient enrollment set to begin in April 2025. In December 2024, Atea announced that its Phase 2 study of bemnifosbuvir and ruzasvir for treating hepatitis C virus (HCV) met its primary endpoints.
Hepatitis C Drug Chapters
The drug chapter segment of the Hepatitis C report encloses a detailed analysis of Hepatitis C marketed drugs and late-stage (Phase III and Phase II) pipeline drugs. It also deep dives into the Hepatitis C pivotal clinical trial details, recent and expected market approvals, patent details, the latest news, and recent deals and collaborations.
Marketed Drugs
MAVYRET (Glecaprevir and Pibrentasvir): AbbVie
MAVYRET (US), MAVIRET (EU, Japan) is a pan-genotypic, once-daily, ribavirin-free , co-formulated next generation HCV treatment including glecaprevir (ABT-493, broad-genotypic NS3/4A protease inhibitor) and pibrentasvir (ABT-530, NS5A inhibitor). Glecaprevir-pibrentasvir is the first pangenotypic NS3/4A protease inhibitor-NS5A inhibitor combination to be approved that offers a potent ribavirin free option for the vast majority of patients with chronic hepatitis C, including a potential eight-week option for non-cirrhotic patients with renal disease or HIV co-infection. This drug is not an option for patients with decompensated cirrhosis given the presence of the protease inhibitor.
VOSEVI (sofosbuvir, velpatasvir, and voxilaprevir): Gilead Sciences
Vosevi is the first pan-genotypic fixed-dose tablet that includes medications from three different HCV antiviral classes, specifically for patients with hepatitis C who were previously treated with certain advanced hepatitis C regimens, but could not achieve a functional cure. It is a prescription medicine used to treat adults with chronic (lasting a long time) hepatitis C genotype with or without cirrhosis (compensated) who have previously been treated with a HCV regimen.
| Therapy Name | Company Name | ROA | MOA | Any Special Status |
| MAVYRET | AbbVie | Oral | HCV NS3/4A protease and NS5B inhibition | Breakthrough Therapy Designation, Priorty Review |
| VOSEVI | Gilead Sciences | Oral | NS5B polymerase inhibitor, HCV NS5A inhibitor, and HCV NS3/4A protease inhibitor | NA |
| EPCLUSA | Gilead Sciences | Oral | HCV nucleotide analog NS5B polymerase inhibitor, and HCV NS5A replication complex inhibitor | NA |
Emerging Drugs
Bemnifosbuvir + Ruzasvir: Atea Pharmaceuticals
Bemnifosbuvir (AT-527) is Atea Pharmaceuticals' purine nucleotide pro-drug platform, which is being developed as an orally administered, direct-acting antiviral therapy for the treatment of patients with HCV. AT-527 is designed to inhibit viral replication by interfering with viral RNA polymerase, a key component in the replication machinery of RNA viruses. Ruzasvir is a NS5A inhibitor. Bemnifosbuvir + Ruzasvir have each separately generate clinical results and are being developed as a comination of HCV. Currently, it is in Phase II of its clinical development.
Note: Detailed therapies assessment will be provided in the final report.
Hepatitis C Market Outlook
The Hepatitis C (HCV) treatment market is on an upward trajectory, fueled by the success of direct-acting antiviral agents (DAAs) with cure rates exceeding 95%. These well-tolerated oral regimens have dramatically improved patient outcomes, replacing older interferon-based therapies. Global efforts, led by the WHO, aim to reduce new infections, driving expanded DAA access, especially in low- and middle-income countries. While prevalence is declining and liver-related complications are decreasing, challenges like reinfection in high-risk groups and equitable treatment access persist.
- In 2023, the United States accounted for approximately USD 2000 million HCV market.
Hepatitis C Drugs Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during 2024–2034, which depends on the competitive landscape, safety, and efficacy data along with order of entry. It is important to understand that the key players evaluating their novel therapies in the pivotal and confirmatory trials should remain vigilant when selecting appropriate comparators to stand the greatest chance of a positive opinion from regulatory bodies, leading to approval, smooth launch, and rapid uptake. Further detailed analysis of emerging therapies drug uptake in the report…
Hepatitis C Activities
The report provides insights into different therapeutic candidates in Phase III and Phase II stages. It also analyzes key players involved in developing targeted therapeutics.
Pipeline Development Activities
The report covers information on collaborations, acquisitions and mergers, licensing, and patent details for Hepatitis C emerging therapies.
KOL Views
To keep up with the real-world scenario in current and emerging market trends, we take opinions from Key Industry leaders working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the evolving treatment landscape, patient reliance on conventional therapies, patient therapy switching acceptability, and drug uptake along with challenges related to accessibility.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 5+ KOLs in the 7MM. Their opinion helps understand and validate current and emerging treatment patterns of Hepatitis C. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
Qualitative Analysis
We perform Qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and Conjoint Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of gaps in disease diagnosis, patient awareness, physician acceptability, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint Analysis analyzes multiple approved and emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
In efficacy, the trial’s primary and secondary outcome measures are evaluated; for instance, in event-free survival, one of the most important primary outcome measures is event-free survival and overall survival.
Further, the therapies’ safety is evaluated wherein the acceptability, tolerability, and adverse events are majorly observed, and it sets a clear understanding of the side effects posed by the drug in the trials. In addition, the scoring is also based on the probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Market Access and Reimbursement
The report provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of currently used therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Scope of the Report
- The report covers a segment of key events, an executive summary, descriptive overview of Hepatitis C, explaining its causes, signs and symptoms, pathogenesis, and currently available therapies.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of diagnosis rate, and disease progression along with country specific treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging therapies, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Hepatitis C market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies, by understanding trends, through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Hepatitis C market.
Hepatitis C Report Insights
- Patient Population
- Therapeutic Approaches
- Hepatitis C Pipeline Analysis
- Hepatitis C Market Size and Trends
- Existing and future Market Opportunity
Hepatitis C Report Key Strengths
- Eleven Years Forecast
- 7MM Coverage
- Hepatitis C Epidemiology Segmentation
- Inclusion of Country specific treatment guidelines
- KOL’s feedback on approved and emerging therapies
- Key Cross Competition
- Conjoint analysis
- Drugs Uptake and Key Market Forecast Assumptions
Hepatitis C Report Assessment
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Market Attractiveness
- Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
- What is the growth rate of the 7MM Hepatitis C treatment market?
- What was the Hepatitis C total market size, the market size by therapies, market share (%) distribution in 2020, and what would it look like in 2034? What are the contributing factors/key catalysts for this growth?
- Is there any unexplored patient setting that can open the window for growth in the future?
- What are the pricing variations among different geographies for approved and off-label therapies?
- How would the market drivers, barriers, and future opportunities affect the market dynamics and subsequent analysis of the associated trends? Although multiple expert guidelines recommend testing for targetable mutations before therapy initiation, why do barriers to testing remain high?
- What are the current and emerging options for the treatment of Hepatitis C ?
- How many companies are developing therapies for the treatment of Hepatitis C ?
- What are the recent novel therapies, targets, mechanisms of action, and technologies developed to overcome the limitations of existing therapies?
- Patient/physician acceptability in terms of preferred treatment options as per real-world scenarios?
- What are the country-specific accessibility issues of expensive, recently approved therapies?
Reasons to buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Hepatitis C Market.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Distribution of historical and current patient share based on real-world prescription data along with reported sales of approved products in the US, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- Highlights of access and reimbursement policies of approved therapies, barriers to accessibility of expensive off-label therapies, and patient assistance programs.
- To understand Key Opinion Leaders’ perspectives around the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.